14.1 POJA-L6293-C Scheme of the skeletal muscle fibre sarcomere model
14.1 POJA-L6293-C Scheme of the skeletal muscle fibre sarcomere model
(Skeletal muscle: A review of molecular structure and function, in health and diseaseKavitha Mukund and Shankar Subramaniam, First published: 13 August 2019, (https://doi.org/10.1002/wsbm.1462). © 2019 The Authors. WIREs Systems Biology and Medicine published by Wiley Periodicals, Inc. This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Title: Scheme of the skeletal muscle fibre sarcomere model
Description:
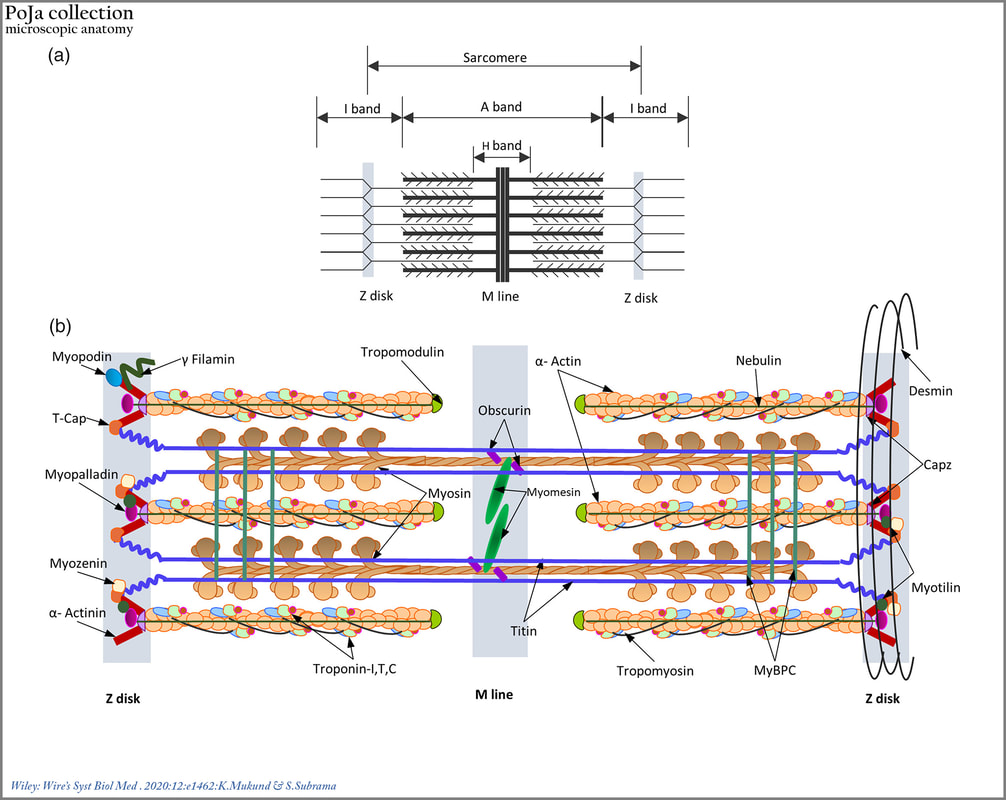
(a): Schematic representation of the striated muscle sarcomere showing the arrangement of thick and thin filaments in the sarcomere and identifying bands of overlap between them.
(b): Schematic diagram of the sarcomere summarizing organization and location of major sarcomeric proteins.
Cytosolic Ca2+ brings about a conformational change in the structure of troponin C, revealing myosin binding sites.
Myosin heads successively bind and crawl along the length of actin, bringing about sarcomeric contraction.
Titin and nebulin, function as “molecular templates” maintaining the length of the thick and thin filaments, respectively.
A whole host of proteins within the M‐line and Z‐disc functions mainly to maintain structural integrity of thick and thin filament lattices, respectively. The desmin intermediate filaments reinforce and integrate the structure of the muscle cell by forming transverse links between adjacent myofibrils.
Histology and molecular structure of the striated muscle.
The muscle fibre contains parallel arranged myofibrils that show light microscopically striations, i.e., alternating thin light and wider dark bands. The dark bands appear strongly birefringent in polarized light and are therefore called anisotropic, A-bands. The light bands are refringent, isotropic, I-bands. Inside the I-bands are the so-called Z-lines (or Z-discs) very narrow anisotropic line. The functional contractive unit between two Z-lines is called sarcomere.
The contractile proteins within the sarcomere are arranged in filaments, one thick filament (myosin) is surrounded by six thin (actin) filaments, creating alternating, overlapping zones of thick and thin filaments (dark and light stained zones).
The dark A-zone is the overlapping zone of myosin and actin, whereas the light I-zone is the zone of thin filaments that does not overlap thick filaments. During muscle contraction the thin filaments of the myofibrils slide over the thick filaments.
One thick filament (myosin) is formed by aggregation of about 300 myosin molecules. The myosin molecule is composed of two tadpole-shaped heavy chains (200 kDa each) (tail portions are coiled around each other) with four small light chains (20 kDa each) attached to the head portions. One myosin head is paired to two myosin light chains. The heavy chain head binds ATP and is called therefore myosin ATPase that splits ATP in ADP and Pi, thus generates the force for the movement along the actin filaments (contraction) by rotating the myosin molecule “neck”, (swinging lever-arm hypothesis). In addition, the heavy chain myosin also has an actin-binding domain. Different myosin isoforms are found in different muscles. (www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/myosin)
The light meromyosin (LMM) fragment of the skeletal myosin II type corresponds to two thirds of the tail and is responsible for the self-assembly of the protein dimers. The heavy meromyosin part (HMM) contains two sub-fragments: the globular head S1 being the motor domain, and the proximal third of the tail, sub-fragment S2, that acts as a flexible connection between S1 and LMM.
The myosin binding protein (MyBP) and the elastic protein titin are associated elements of the thick filaments.
Thin filaments are mainly composed of protein actin (42 kDa). Each thin filament (F-actin) is formed by polymerization of single globular actin (G-actin) molecules. Two actin filaments become attached by their tail ends to α-actinin in the Z-line. The F-actin chains are stabilized by complex formation with tropomyosin and troponin. The troponin consists of three subunits: troponin I (TnI) binds to actin; troponin T (TnT) binds to tropomyosin; troponin C (TnC) binds to Ca ions.
Accessory proteins link the different components and hold them stabilized with each other.
Keywords/Mesh: locomotor system, skeletal muscle, striated muscle, sarcomere, costamere, myofilament, myosin, actin, A-band, I-band, H-band, M-band, Z-line, cytoskeleton, scheme, electron microscopy, POJA collection
Title: Scheme of the skeletal muscle fibre sarcomere model
Description:
(a): Schematic representation of the striated muscle sarcomere showing the arrangement of thick and thin filaments in the sarcomere and identifying bands of overlap between them.
(b): Schematic diagram of the sarcomere summarizing organization and location of major sarcomeric proteins.
Cytosolic Ca2+ brings about a conformational change in the structure of troponin C, revealing myosin binding sites.
Myosin heads successively bind and crawl along the length of actin, bringing about sarcomeric contraction.
Titin and nebulin, function as “molecular templates” maintaining the length of the thick and thin filaments, respectively.
A whole host of proteins within the M‐line and Z‐disc functions mainly to maintain structural integrity of thick and thin filament lattices, respectively. The desmin intermediate filaments reinforce and integrate the structure of the muscle cell by forming transverse links between adjacent myofibrils.
Histology and molecular structure of the striated muscle.
The muscle fibre contains parallel arranged myofibrils that show light microscopically striations, i.e., alternating thin light and wider dark bands. The dark bands appear strongly birefringent in polarized light and are therefore called anisotropic, A-bands. The light bands are refringent, isotropic, I-bands. Inside the I-bands are the so-called Z-lines (or Z-discs) very narrow anisotropic line. The functional contractive unit between two Z-lines is called sarcomere.
The contractile proteins within the sarcomere are arranged in filaments, one thick filament (myosin) is surrounded by six thin (actin) filaments, creating alternating, overlapping zones of thick and thin filaments (dark and light stained zones).
The dark A-zone is the overlapping zone of myosin and actin, whereas the light I-zone is the zone of thin filaments that does not overlap thick filaments. During muscle contraction the thin filaments of the myofibrils slide over the thick filaments.
One thick filament (myosin) is formed by aggregation of about 300 myosin molecules. The myosin molecule is composed of two tadpole-shaped heavy chains (200 kDa each) (tail portions are coiled around each other) with four small light chains (20 kDa each) attached to the head portions. One myosin head is paired to two myosin light chains. The heavy chain head binds ATP and is called therefore myosin ATPase that splits ATP in ADP and Pi, thus generates the force for the movement along the actin filaments (contraction) by rotating the myosin molecule “neck”, (swinging lever-arm hypothesis). In addition, the heavy chain myosin also has an actin-binding domain. Different myosin isoforms are found in different muscles. (www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/myosin)
The light meromyosin (LMM) fragment of the skeletal myosin II type corresponds to two thirds of the tail and is responsible for the self-assembly of the protein dimers. The heavy meromyosin part (HMM) contains two sub-fragments: the globular head S1 being the motor domain, and the proximal third of the tail, sub-fragment S2, that acts as a flexible connection between S1 and LMM.
The myosin binding protein (MyBP) and the elastic protein titin are associated elements of the thick filaments.
Thin filaments are mainly composed of protein actin (42 kDa). Each thin filament (F-actin) is formed by polymerization of single globular actin (G-actin) molecules. Two actin filaments become attached by their tail ends to α-actinin in the Z-line. The F-actin chains are stabilized by complex formation with tropomyosin and troponin. The troponin consists of three subunits: troponin I (TnI) binds to actin; troponin T (TnT) binds to tropomyosin; troponin C (TnC) binds to Ca ions.
Accessory proteins link the different components and hold them stabilized with each other.
- α-Actinin: links and stabilizes the actin filaments in the Z-disc.
- Myomesin: (MYOM1, 2, 3) holds myosin filaments in a lattice arrangement in the region of the M line. They are structural linkers between the thick filaments.
- Titin: Is a giant muscular protein (3MDa). Each titin molecule expands along the sarcomere from the Z-line to the M-line. It provides the passive mechanical tension necessary for the sarcomere to restore its resting length after contraction. It is considered as a molecular spring between the Z-line and the M-line. The titin molecule has binding domains for α-actinin and telethonin.
- Desmin: an intermediate filament (53 kDa) that links adjacent myofibrils to each other as well as to the costameres and cell membrane in the extra-myofibrillar space.
- Tropomyosin: Is a long rod-like protein that winds around an actin filament to stabilize and stiffen it. A molecule of 37 kDa that forms homo- or heterodimers of chains and helical structure all over 7 actin subunits. The primary function is to regulate the muscle contraction by blocking the binding sites of myosin located in actin protein. Thus, it prevents the actin-myosin interaction in relaxed state.
- Troponin complex: regulates the binding of actin to myosin. It is attached to tropomyosin and consists of 3 subunits:
- troponin C (TnC, 18 kDa) that binds Ca ions which cause a conformational change in the troponin complex allowing myosin access to the actin filaments;
- troponin I (TnI, 20-24 kDa) physically prevents myosin binding to actin when complexed with actin at low Ca ions concentration;
- troponin T (TnT, 31-36 kDa) binds the complex to tropomyosin and positions the complex on the actin filament. One troponin complex is present for every seven actin molecules along the thin filaments.
- Nebulin: a giant protein (600-900 kDa) with highly repetitive structure of units of 30-35 amino acids. A single molecule of nebulin extends along each thin filament with its COOH terminal anchored to the Z-line and the NH2 terminal facing the other site of the filament. Nebulin interacts also with desmin maintaining the lateral alignment of myofibrils.
- Synemin and nestin: The synemin molecule (230 kDa) is located at the Z-line and co-polymerizes with desmin protein. It has binding sites for α-actinin and vinculin and is involved in the connection between Z-line and cell membrane. Nestin (220-240 kDa) is coexpressed temporarily with vimentin and desmin on the Z-line during prenatal development.
- Syncoilin: (64 kDa) interacts with desmin and with the dystrophin-associated protein complex (DGC). Syncoilin is expressed at the neuromuscular junction, the sarcolemma and the Z-line. It interacts with the intermediate filaments network anchoring to the sarcolemma.
- Skelemin: (195 kDa) protein located on the M line and it mediates the connection to the extracellular matrix in early stages of the development
- Plectin: (>500 kDa) links intermediate filaments and costameres in the Z-line area. It also anchors organelles (mitochondria) and nuclei to the intermediate filament network.
- Obscurin: A protein that mediates links between the M-band and the sarcoplasmic reticulum as well as the plasma membrane.
- The costamere comprises several more proteins to link sarcomere filaments with the sarcolemma, as illustrated in this figure.
- For detailed description of these and other proteins associated with the sarcomere such as myopodin, filamin, myopalladin, myozenin, tropomodulin, myotilin, obscurin see the review article by K. Mukund & S. Subramaniam (https://doi.org/10.1002/wsbm.1462): Skeletal muscle: A review of molecular structure and function, in health and disease
Keywords/Mesh: locomotor system, skeletal muscle, striated muscle, sarcomere, costamere, myofilament, myosin, actin, A-band, I-band, H-band, M-band, Z-line, cytoskeleton, scheme, electron microscopy, POJA collection